Penicillium
Penicillium is a very large and ubiquitous genus which currently contains 354 accepted species (Visagie et al. 2014).
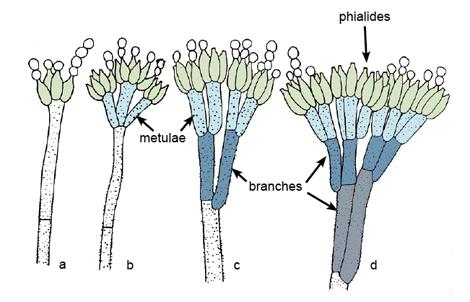
Morphological structures and types of conidiophore branching in Penicillium (a) Monoverticillate; (b) Biverticillate; (c) Terverticillate; (d) Quaterverticillate (see Visagie et al. 2014).
Many species are common contaminants on various substrates and are known as potential mycotoxin producers. Correct identification is therefore important when studying possible Penicillium contamination of food. Human pathogenic species are rare, however opportunistic infections leading to mycotic keratitis, otomycosis and endocarditis (following insertion of valve prosthesis) have been reported (Lyratzopoulos et al. 2002). Note: Penicillium marneffei and other subgenus Biverticillium species have been transferred to the genus Talaromyces (Samson et al. 2011b).
RG-1 organisms.
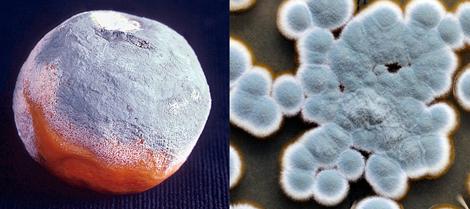
Culture of Penicillium spp.
Morphological description:
Colonies are usually fast growing, in shades of green, sometimes white, mostly consisting of a dense felt of conidiophores. Microscopically, chains of single-celled conidia are produced in basipetal succession from a specialised conidiogenous cell called a phialide. The term basocatenate is often used to describe such chains of conidia where the youngest conidium is at the basal or proximal end of the chain. In Penicillium, phialides may be produced singly, in groups or from branched metulae, giving a brush-like appearance (a penicillus). The penicillus may contain both branches and metulae (penultimate branches which bear a whorl of phialides). All cells between the metulae and the stipes of the conidiophores are referred to as branches. The branching pattern may be either simple (non-branched or monoverticillate), one-stage branched (biverticillate-symmetrical), two-stage branched (biverticillate-asymmetrical) or three- to more-staged branched. Conidiophores are hyaline, smooth or rough-walled. Phialides are usually flask-shaped, consisting of a cylindrical basal part and a distinct neck, or lanceolate (with a narrow basal part tapering to a somewhat pointed apex). Conidia are in long dry chains, divergent or in columns, are globose, ellipsoidal, cylindrical or fusiform, hyaline or greenish, smooth or rough-walled. Sclerotia are produced by some species.
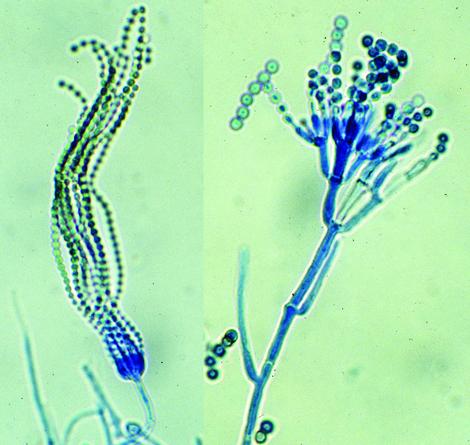
(Left) Simple conidiophore of P. cheresanum showing long chains of single-celled phialoconidia and (right) conidiophores of P. verrucosum var. cyclopium showing two-stage branching.
For identification, isolates are usually inoculated at three points on Czapek Dox agar and 2% Malt extract agar and incubated at 25C. Most species sporulate within 7 days. Microscopic mounts are best made using a cellotape flag or a slide culture preparation mounted in lactophenol cotton blue. A drop of alcohol is usually needed to remove bubbles and excess conidia (Samson et al. 1995).
Molecular identification:
ITS and/or β-tubulin loci are recommended for identification of Penicillium species (Visagie et al. 2014, Yilmaz et al. 2014).
Key features:
Hyphomycete, flask-shaped phialides arranged in groups from branched metulae forming a penicillus.
References:
Raper and Thom (1949), Pitt (1979), Domsch et al. (1980), McGinnis (1980), Onions et al. (1981), Ramirez (1982), Samson et al. (1995, 2011b), de Hoog et al. (2000, 2015), Visagie et al. (2014).
|
Antifungal susceptibility: Penicillium spp. (Australian national data); MIC µg/mL. |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| No | ≤0.03 | 0.06 | 0.125 | 0.25 | 0.5 | 1 | 2 | 4 | ≥8 | |
| AmB | 88 | 4 | 6 | 12 | 17 | 27 | 15 | 7 | ||
| ISAV | 33 | 3 | 3 | 6 | 9 | 5 | 6 | 1 | ||
| VORI | 86 | 1 | 5 | 7 | 14 | 13 | 15 | 5 | 26 | |
| POSA | 86 | 4 | 7 | 21 | 16 | 25 | 7 | 1 | 5 | |
| ITRA | 88 | 2 | 3 | 16 | 16 | 29 | 13 | 3 | 6 | |
