Apophysomyces complex
Historically the genus Apophysomyces was considered to be monotypic and A. elegans was reported to be an important human pathogen in immunocompetent patients following traumatic implantation.
A phylogenetic revision of the genus has identified three additional species, A. mexicanus, A. ossiformis, A. trapeziformis and A. variabilis. Many isolates previously identified as A. elegans, now appear to be A. variabilis (Alvarez et al. 2010a). Interestingly, most of the human clinical isolates tested appear to be A. variabilis and not A. elegans as previously reported (Alvarez et al., 2010a; Walther et al., 2019). Molecular identification is required to accurately differentiate these species. Morphological characteristics overlap so identification and reporting of Apophysomyces complex is recommended for most diagnostic laboratories.
Molecular identification:
The ITS region and D1/D2 domain may provide for accurate species identification (Halliday et al. 2015). ITS restriction fragment length polymorphism analysis has also been described (Chakrabarti et al. 2003).
Apophysomyces elegans
RG-2 organism.
Culture of Apophysomyces elegans.
Morphological description: Colonies are fast growing, white, becoming brownish grey with age, downy with no reverse pigment, and are composed of broad, sparsely septate (coenocytic) hyphae. Sporangiophores are unbranched, straight or curved, slightly tapering towards the apex, up to 540 µm long, 3-6 µm in width near the apophysis, and hyaline when young but developing a light to dark brown pigmentation and a conspicuous sub-apical thickening 10-16 µm below the apophysis with age. Sporangiophores arise at right angles from the aerial hyphae and often have a septate basal segment resembling the “foot cell” commonly seen in Aspergillus. Rhizoids are thin-walled, subhyaline and predominantly unbranched. Sporangia are multispored, small (20-58 µm diameter), typically pyriform in shape, hyaline at first, sepia coloured when mature, columellate and strongly apophysate. Columellae are hemispherical in shape and the apophyses are distinctively funnel or bell-shaped. Sporangiospores are smooth-walled, subspherical to cylindrical, (5-8 x 4-6 µm), subhyaline to sepia in mass. Good growth at 26C, 37C and 42C.
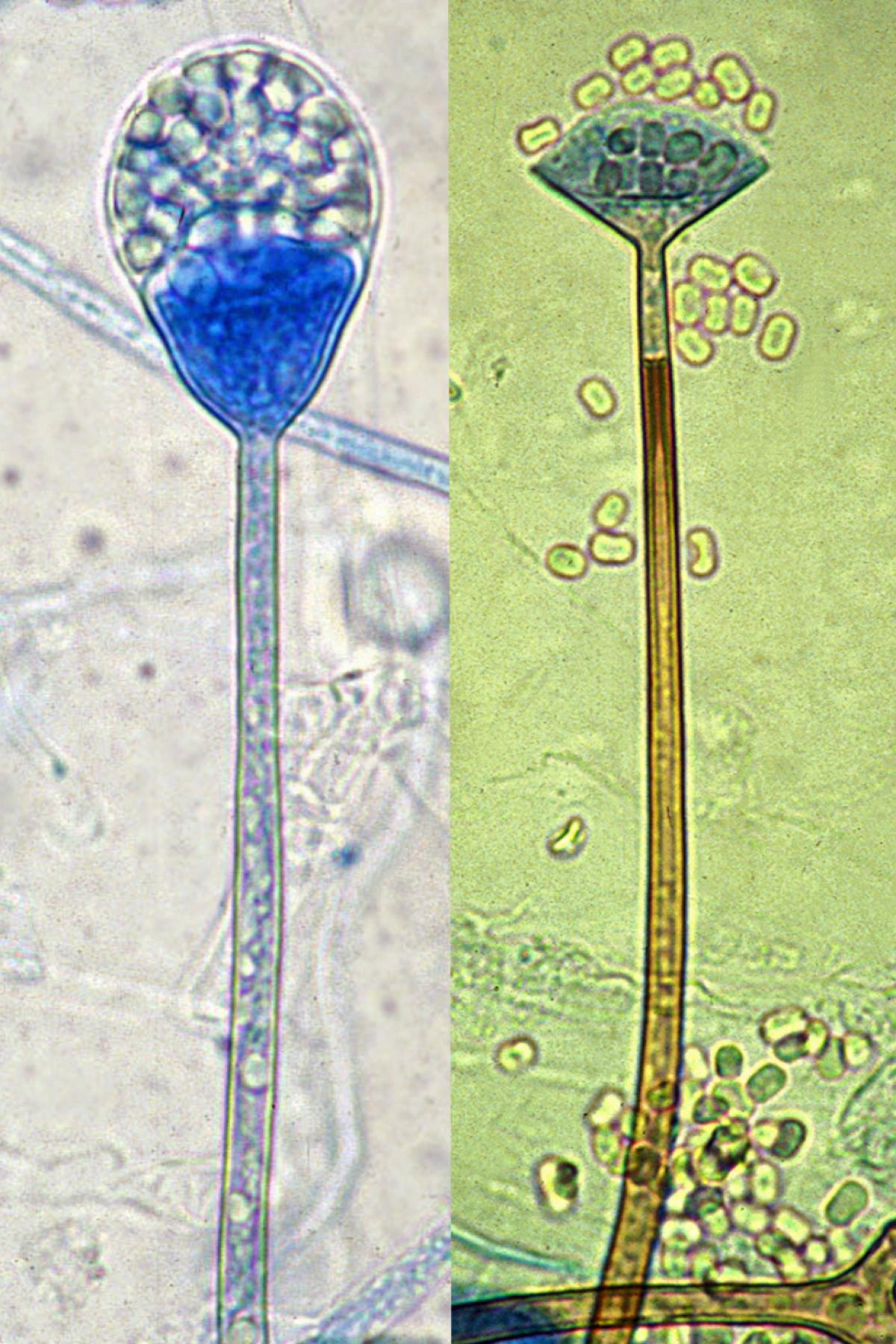
Young sporangium (left) and mature sporangium (right) of Apophysomyces elegans showing distinctive funnel-shaped apophyses and a conspicuous pigmented sub-apical thickening below the apophysis.
Key features:
Soil fungus with a tropical to subtropical distribution. Characteristic “cocktail glass” apophysate sporangial morphology with conspicuous subapical thickening of the sporangiophore. Resistance to cycloheximide. Rapid growth at 42C, no growth at 50C.
Comment:
Apophysomyces complex is readily distinguished from other zygomycetes, especially the morphologically similar, strongly apophysate pathogen Lichtheimia corymbifera, by having sporangiophores with distinctive funnel or bell-shaped apophyses and hemispherical-shaped columellae. In addition, there is a conspicuous pigmented subapical thickening, which constricts the lumen of the sporangiophore below the apophysis, and distinctive foot cells.
Note:
Laboratory identification of this fungus may be difficult or delayed because of the mould’s failure to sporulate on primary isolation media or on subsequent subculture onto potato dextrose agar. Sporulation may be stimulated by the use of nutrient deficient media, like cornmeal-glucose-sucrose-yeast extract agar, Czapek Dox agar, or by using the agar block method described by Ellis and Ajello (1982) and Ellis and Kaminski (1985). Molecular-based identification is particularly helpful for the definitive identification of poorly sporulating cultures.
| Antifungal | No | 0.016 | 0.03 | 0.06 | 0.125 | 0.25 | 0.5 | 1 | 2 | 4 | ≥8 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| AmB | 24 | 3 | 7 | 2 | 12 | ||||||
| ISAV | 20 | 2 | 10 | 8 | |||||||
| VORI | 26 | 2 | 24 | ||||||||
| POSA | 24 | 4 | 7 | 10 | 3 | ||||||
| ITRA | 26 | 1 | 7 | 12 | 6 |
Apophysomyces variabilis
RG-2 organism.
Morphological Description:
Colonies are fast growing, whitish with scarce aerial mycelium and no reverse pigment. Sporangiophores are erect, generally arising singly, unbranched, slightly tapering towards the apex, up to 100-400 µm long, 2-3.5 µm in width near the apophysis, hyaline when young but developing a light greyish brown pigmentation with age. Sporangia are multispored, small (15-50 µm diameter), typically pyriform in shape, hyaline at first, sepia-coloured when mature, with distinct apophyses and columellae. Columellae are hemispherical in shape and the apophyses are short and distinctly funnel-shaped. Sporangiospores are smooth-walled, variable in shape, trapezoid, ellipsoidal, sub-triangular or claviform, (5-14 x 3-6 µm), subhyaline to sepia in mass. Good growth at 26C, 37C and 42C.
References:
Misra et al. (1979), Ellis and Ajello (1982), Padhye and Ajello (1988), Wieden et al. (1985), Lawrence et al. (1986), Cooter et al. (1990), Holland (1997), de Hoog et al. (2000, 2015), Ellis (2005b), Alvarez et al. (2010a), Chakrabarti et al.(2003, 2010), Guarro et al. (2011).
| Antifungal | No | 0.016 | 0.03 | 0.06 | 0.125 | 0.25 | 0.5 | 1 | 2 | 4 | 8 | ≥16 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AmB | 13 | 1 | 1 | 1 | 5 | 5 | ||||||
| ISAV | 3 | 2 | 1 | |||||||||
| VORI | 3 | 3 | ||||||||||
| POSA | 13 | 2 | 2 | 8 | 1 | |||||||
| ITRA | 3 | 3 |
| Antifungal | Range | MIC90 | Antifungal | Range | MIC90 |
|---|---|---|---|---|---|
| AMB | 0.125-4 | 4 | VORI | 16-≥16 | >16 |
| POSA | 0.03-2 | 1 | ITRA | 0.03-2 | 1 |
